Cesium carbonate CAS#534-17-8
CAS Number: 534-17-8
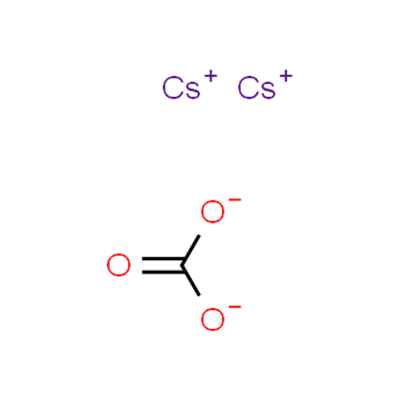
Chemical Formula: CCs2O3
Synonyms:
CAESIUM CARBONATE
Cesium carbonate, for analysis
Carbonic acid dicesium
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: White Powder
Cesium carbonate CAS#534-17-8
Cesium carbonate is an inorganic compound. It is a white solid under normal temperature and pressure. It is easily soluble in water and quickly absorbs moisture when placed in the air. Cesium carbonate aqueous solution is strongly alkaline and can react with acid to produce corresponding cesium salt and water, and release carbon dioxide. Cesium carbonate is easy to transform, can be used as a precursor of other cesium salts, and is a widely used cesium salt.
Cesium carbonate Chemical Properties |
Melting point | 610 °C (dec.) (lit.) |
density | 4.072 |
vapor pressure | 0Pa at 25℃ |
storage temp. | Inert atmosphere,Room Temperature |
solubility | soluble in Water |
form | Powder/Granules |
Specific Gravity | 4.072 |
color | White |
PH | 10-12 (20°C, 50g/L in H2O) |
Water Solubility | 261 g/100 mL (20 ºC) |
Sensitive | Hygroscopic |
Merck | 14,2010 |
BRN | 4546405 |
Stability: | Stable. Very deliquescent. Incompatible with strong oxidizing agents, strong acids. |
InChI | 1S/CH2O3.2Cs/c2-1(3)4;;/h(H2,2,3,4);;/q;2*+1/p-2 |
InChIKey | FJDQFPXHSGXQBY-UHFFFAOYSA-L |
SMILES | [Cs+].[Cs+].[O-]C([O-])=O |
LogP | 0 at 20℃ |
CAS DataBase Reference | 534-17-8(CAS DataBase Reference) |
EPA Substance Registry System | Carbonic acid, dicesium salt (534-17-8) |
Safety Information |
Hazard Codes | Xn,Xi |
Risk Statements | 68-36/37/38 |
Safety Statements | 22-24/25-36/37/39-26-27 |
WGK Germany | 2 |
RTECS | FK9400000 |
F | 3-10 |
TSCA | TSCA listed |
HS Code | 28369918 |
Storage Class | 13 - Non Combustible Solids |
Hazard Classifications | Eye Dam. 1 |
Product Application Of Cesium carbonate CAS#534-17-8
Cesium carbonate is widely utilized as a precursor for other cesium compounds. It acts as a base in sensitive organic reactions. It can be used as a base in C-C and C-N cross-coupling reactions such as SuzukiMiyaura, Heck, and Buchwald-Hartwig amination reactions. It finds use in solar cells as it increases the power conversion efficiency of cells through the transfer of electrons. It is also used in the production of special optical glasses, petroleum catalytic additives, special ceramics and in the sulfuric acid industry. It is useful in the N-alkylation (of sulfonamides, beta-lactams, indoles, heterocycles and several sensitive nitrogen compounds), carbamination of amines, carbonylation of alcohols and aerobic oxidation of alcohols into carbonyl compounds without polymeric by products. Promotes the efficient O-alkylation of alcohols to form mixed alkyl carbonates. |
Fact Factory and Equipment Show
Fast delivery time
Inventory 2-3 working days New production 7-10 working days